Medical Registers
Your one-stop shop for medical quality registers
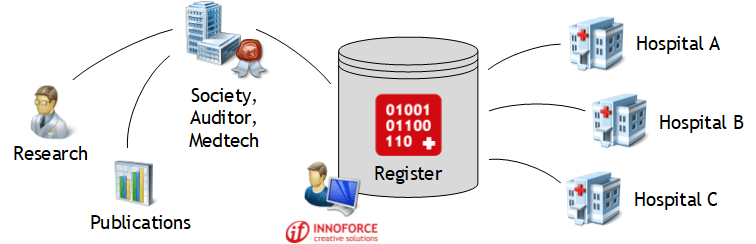
Benefit from the extensive know-how INNOFORCE has acquired over many years in the development and operation of medical quality registers.
Thanks to the tailor-made complete solutions available as a carefree package, you can fully concentrate on your core business. INNOFORCE guarantees you a high-quality concept development, project implementation, system introduction with user training as well as the flawless operation of the register with competent customer support (hotline). INNOFORCE acts as an independent and neutral partner towards all stakeholders.
The INNOFORCE online registers are characterised by a high degree of usability and offer users decisive added value:
- Simple and rapid recording of important therapy parameters
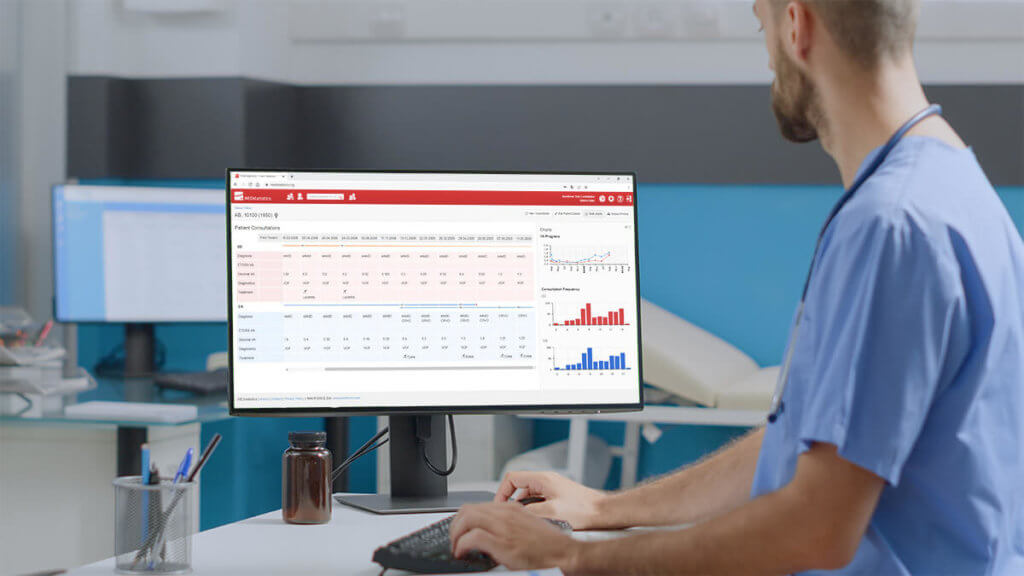
- Clear visualisation of the therapy progress (graphical and tabular)
- Extensive filtering and statistical data analysis
- Excel export for further analyses
- Secure storage and processing of sensitive data in compliance with the GDPR in a state-of-the-art data centre
This added value for the individual user contributes decisively to a high level of compliance in data entry and thus to the success of a register project.
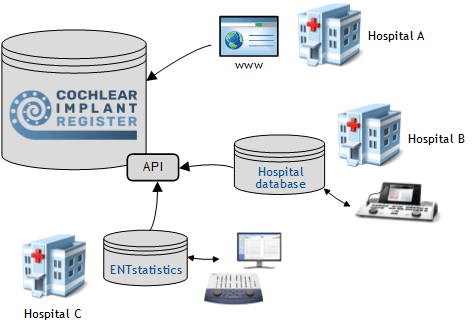
In addition to direct entry via the website portal, electronic import interfaces (API) are also available. This allows data from existing systems to be automatically transferred to the register. There is no need for duplicate data entry: this saves time and contributes to gapless data collection.
Reference projects
DGHNO CI Register
One reference project is the development and operation of the DGHNO Cochlear Implant Register. In addition to manual entry via the website portal, this register also supports data import via an electronic interface (API). This allows therapy data to be transferred to the registry from a clinic-internal database such as ENTstatistics and saves a large amount of time and costs.
Retina Registry for the Swiss Federal Office of Public Health (FOPH)
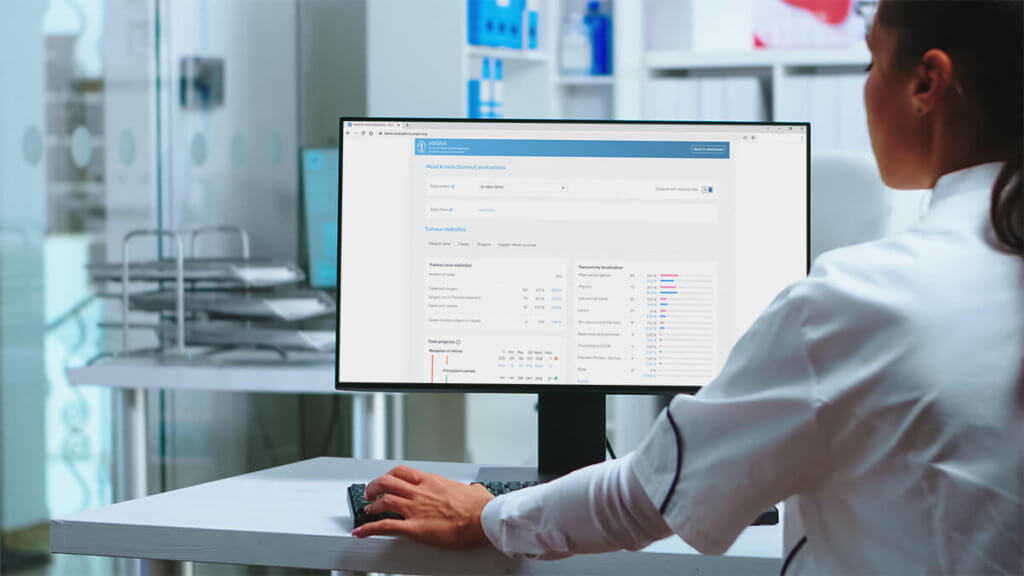
The FOPH Retina Registry was created to collect and statistically analyse data on intravitreal ocular medication treatments (IVOM). The register visualises the course of therapy of each patient and offers users significant added value thanks to numerous evaluations.
DGHNO CI-Register
One reference project is the development and operation of the DGHNO Cochlear Implant Register. In addition to manual entry via the website portal, this register also supports data import via an electronic interface (API). This allows therapy data to be transferred to the registry from a clinic-internal database such as ENTstatistics and saves a large amount of time and costs.
Retina Registry for the Swiss Federal Office of Public Health (FOPH)
The FOPH Retina Registry was created to collect and statistically analyse data on intravitreal ocular medication treatments (IVOM). The register visualises the course of therapy of each patient and offers users significant added value thanks to numerous evaluations.
Australian Otolaryngology Quality Assurance Network (AOQN)
The Australian Otolaryngology Quality Assurance Network (AOQAN) is the national quality register of the Australian Society of Otolaryngology Head and Neck Surgery. It includes modules for cochlear implants, rhinology, tumours, adenoids, tonsils and grommets.
References
The presentation of the FOPH Retina Registry was so well received by senior management that the Head of Marketing Speciality asked me if I would recommend innoForce. Responded in the positive, drawing attention to our prompt and reliable collaboration, your competence, your approach,..., and the positive Vendor Assessment."

Rotkreuz, Switzerland
This is a huge leap forward for all patients and practicing surgeons. I urge all surgeons to embrace this program."

Brisbane, Australia

Berne, Switzerland
Publications
- Baljic, I., Editorial – Z Audiol 2025; 64 (1) 5, PDF
- Wille, C., Qualitätssicherung in der Patientenversorgung vor 100 Jahren und heute – Z Audiol 2025; 64 (1) 19–21, PDF
- Stöver, T., Plontke, S.K., Lai, W.K. et al. The German cochlear implant registry: one year experience and first results on demographic data. European Archives of Oto-Rhino-Laryngology (2024). https://doi.org/10.1007/s00405-024-08775-x
- Stöver, T., Plontke, S.K., Guntinas-Lichius, O. et al. Structure and establishment of the German Cochlear Implant Registry (DCIR). HNO 71 (Suppl 1), 82–92 (2023). https://doi.org/10.1007/s00106-023-01310-0
- Stöver, T., Plontke, S.K., Guntinas-Lichius, O. et al. Conception and implementation of a certification system for quality control of cochlear implant treatment in Germany. HNO 71 (Suppl 1), 73–81 (2023). https://doi.org/10.1007/s00106-023-01306-w
- DGHNO. (2022). HNO-Fachgesellschaft zertifiziert Cochlea-Implantat-Versorgung: Diagnose, Therapie und Nachsorge in guten Händen. Pressemitteilung.
- Coman, W. B., Fisher, P., & Dawes, P. (2021). Surgical outcomes: a 100 years in the waiting – Otolanrygology Head and Neck Surgery pan-national audit. ANZ journal of surgery, 91(9), 1658–1659.
A solution for
your needs?
Are you looking for a solution for your specialist area? Benefit from our know-how from numerous, successfully implemented register projects. We look forward to hearing from you.